 This new model is referred to as the Plum Pudding Model. The New Model of the Atom- Thompson knew atoms were neutral, so there must be a balance of negative and positive particles. The electron is discovered, J J Thomson publishes his discovery of a subatomic particle common to all matter. This was found to be constant regardless of the gas used in the tube and the metal of the cathode and was approximately 1000 times less than the value calculated for hydrogen ions in the electrolysis of liquids. In this section I will discuss the grounds for belief in the existence of the electron by examining J.J. Researchers trying to understand this phenomenon found that an unusual form of energy was also emitted from the cathode, or negatively charged electrode this form of energy was called a cathode ray. 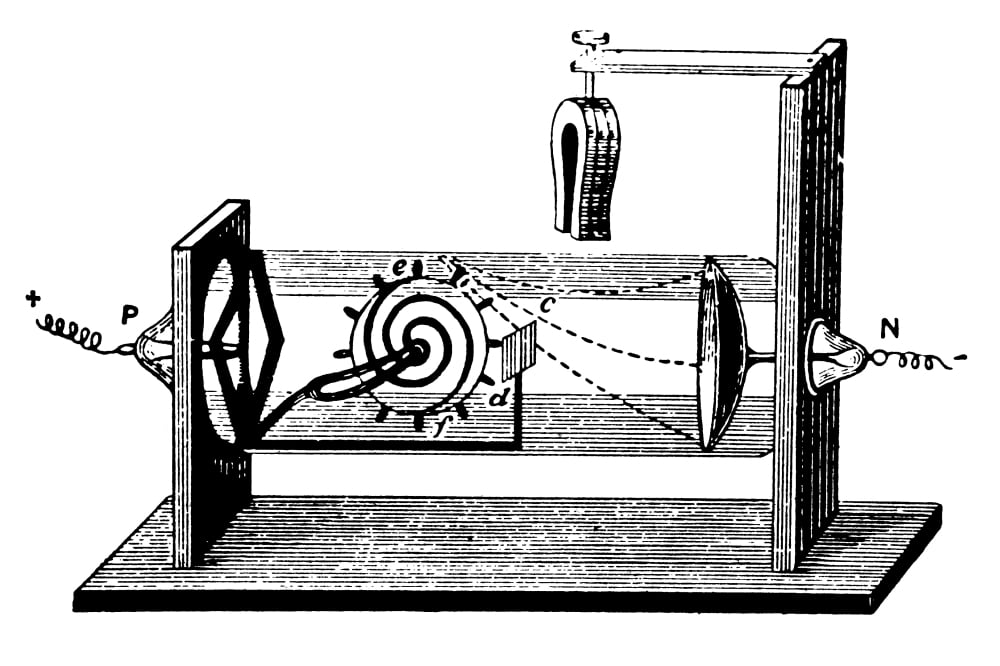
When investigating cathode rays using a highly evacuated discharge tube he was able to use the calculated velocity and deflection of the beam to calculate the ratio of electric charge to mass of the cathode ray. 
Amber when rubbed builds up static charge. Another British electrochemist John Stoney called it the "electron" from the Greek term "Elecktra" which is the Greek word for amber. He called the electron, the "corpuscle" of electrical charge. Thomson, an English physicist, began a series of experiments that would resolve the controversy about cathode rays and lead to the discovery. Thomson and the Cavendish Laboratory in his day. 
Thomson-Professor of Physics and Director of Cavendish Lab at Cambridge UniversityĮlectrons are distributed throughout uniformly charged positive sphere of atomic dimensions. In a series of experiments using cathode ray tubes, Thomson concluded that cathode rays. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
